
James H. Clark Center, Stanford University

James H. Clark Center, Stanford University
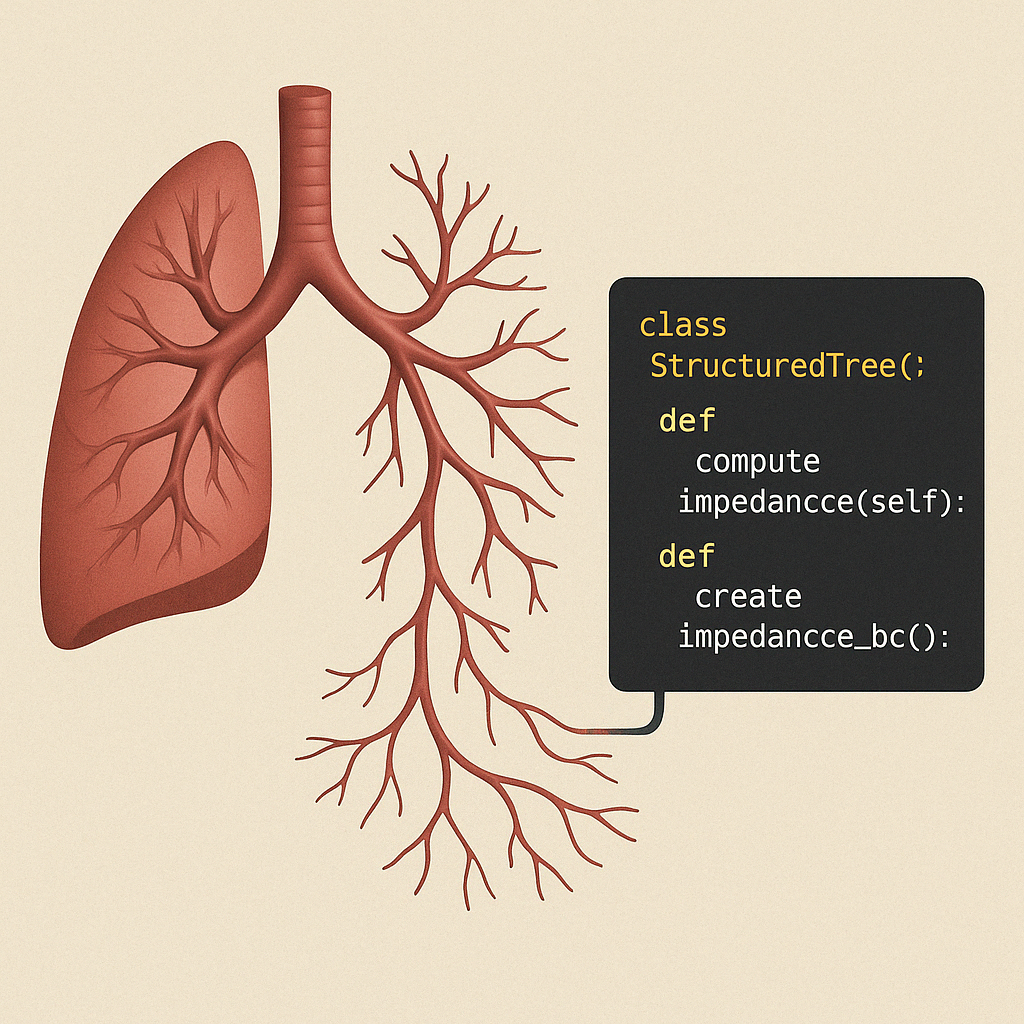
This project involves developing computational models to simulate how microvascular networks adapt to physiological and pathological conditions. The goal is to understand the regulatory mechanisms involved in microvascular growth and remodeling in response to stimuli such as blood flow and pressure.
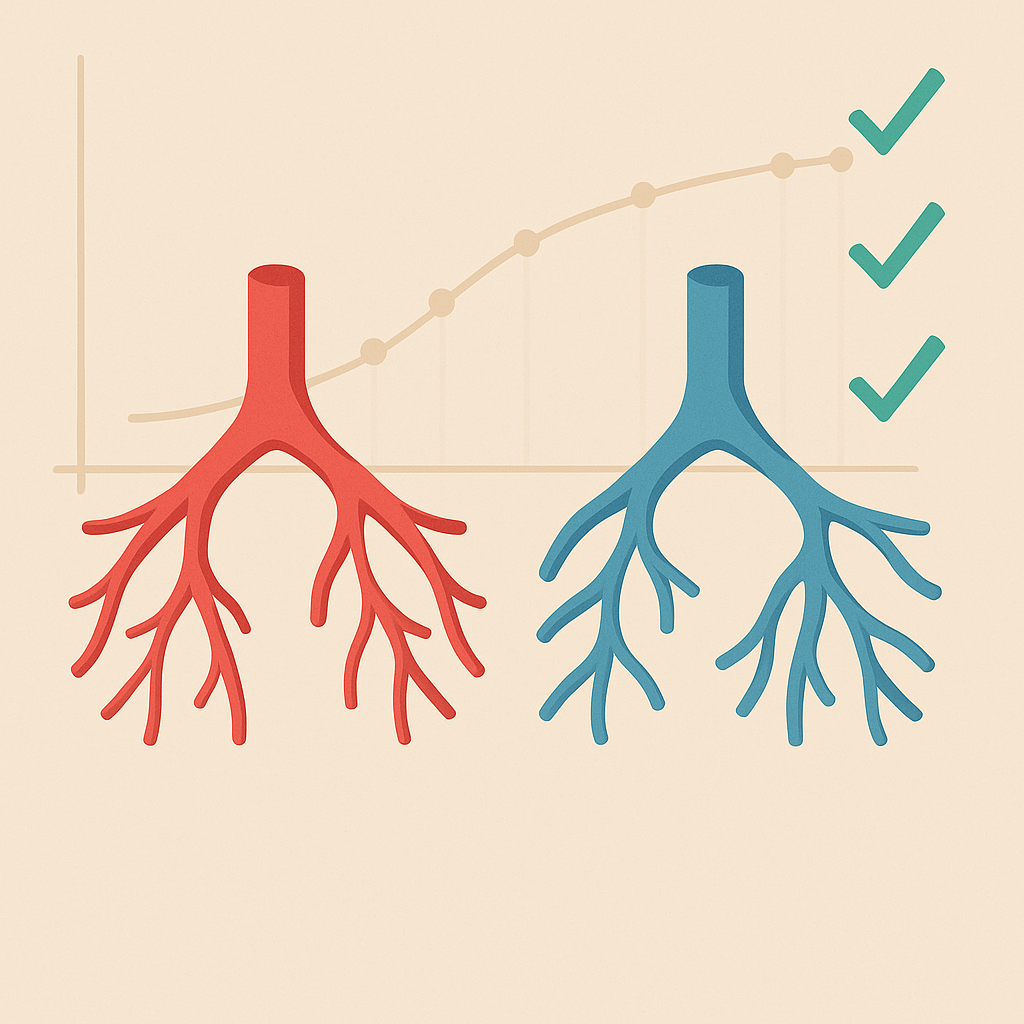
In collaboration with clinical teams, this project focuses on validating the developed microvascular adaptation model in patients with Tetralogy of Fallot (ToF) and Alagille syndrome. The study involves comparing simulation results with patient-specific data to assess the accuracy and predictive capacity of the model.
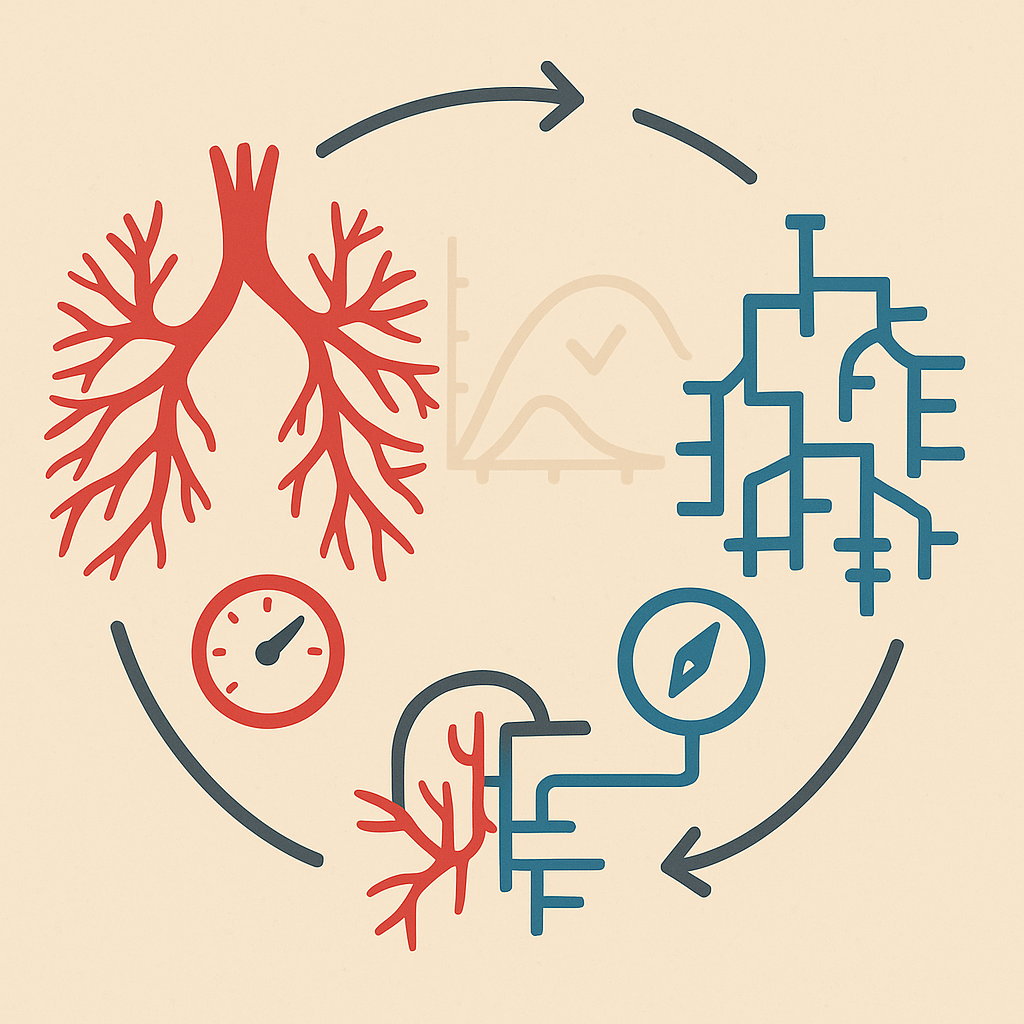
This research aims to create a multi-fidelity closed loop model of the pulmonary arteries that integrates both high-fidelity local models and lower-fidelity global models. The model will be used to study blood flow dynamics and arterial pressure regulation in different pathological scenarios.

I contribute to the development of svZeroDSolver, an open-source solver for reduced-order models of cardiovascular blood flow. This tool enables efficient simulation of complex hemodynamic systems using 0D lumped parameter networks, and is a core component of the SimVascular platform for cardiovascular biomechanics research.
Find Nicholas Dorn’s research publications and contributions on Google Scholar: